
Nature Nanotechnology Article
I. Research Background: Limitations of Traditional Catalysts and New Opportunities for Liquid Metals
Traditional methane oxidation catalysts are primarily based on solid materials, where the chemical environment of active sites determines the catalyst's activity and selectivity. Single-atom catalysts typically produce only C1 products, while clusters or particles with multiple atomic sites usually generate C2 or longer-chain hydrocarbons. However, these solid catalysts have an inherent limitation: their rigid solid structure restricts the ability to modify the chemical configuration of active sites during the reaction process, which significantly limits the possibilities for adjusting catalytic activity and selectivity.
Liquid metals, as emerging materials, provide a new approach for developing methane oxidation catalysts. Their unique physical and chemical properties, such as low melting point, high thermal conductivity, and excellent chemical stability, make them ideal catalyst supports. Compared to solid catalysts, liquid metals remain liquid at room temperature, and atoms are not constrained by crystal lattices, thus providing the possibility of dynamically tunable structures. Unlike traditional catalytic regulation methods, magnetic fields can be easily generated by permanent magnets and allow external control of catalytic reactions under milder conditions.
II. Research Content: Design and Performance Validation of Fe-LMS Catalyst
(A) Catalyst Synthesis and Structural Characterization
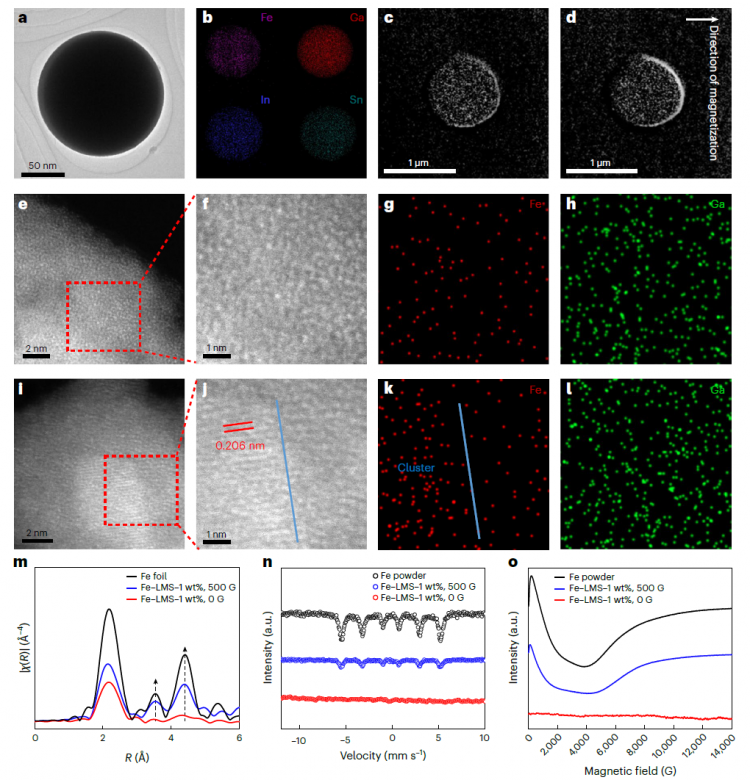
Figure 1. Fe-LMS Structural Characterization
The research team designed an iron-embedded liquid metal catalyst (Fe-LMS), using the eutectic alloy galinstan as a weakly bound substrate. This Fe-LMS catalyst responds to changes in external magnetic field intensity, enabling reversible and rapid rearrangement of iron atoms between isolated single atoms (Fe1-LMS) and aggregated clusters (Fen-LMS).
Through advanced characterization techniques including high-angle annular dark-field scanning transmission electron microscopy (HAADF-STEM), scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS), and synchrotron radiation X-ray 3D imaging reconstruction, the researchers conducted in-depth analysis of the catalyst structure. Under 0 G magnetic field conditions, the sample showed amorphous dispersion of iron/gallium-indium-tin alloy elements. In contrast, a 500 G magnetic field induced the formation of α-Fe(100) clusters (lattice spacing 0.206 nm), while gallium remained uniformly distributed. X-ray absorption fine structure (XAFS) analysis revealed magnetic field-induced structural reorganization.
(B) Magnetic Field-Controlled Methane Oxidation Reaction
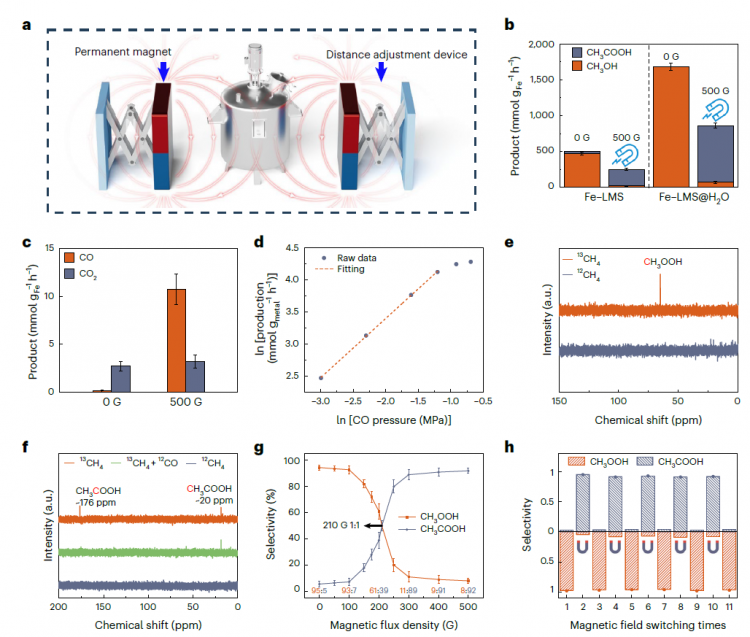
Figure 2. Methane Oxidation Catalytic Performance
The methane oxidation reaction was conducted in a high-pressure autoclave using Fe-LMS droplet catalysts and diluted H2O2. In the absence of a magnetic field, Fe-LMS primarily produced CH3OOH (471.1 mmol·g-1Fe·h-1, 94.5%). At 500 G, the product selectivity reversed, with CH3COOH becoming the main product (230.6 mmol·g-1Fe·h-1, 91.9%). Ultrasonic dispersion generated 100 nm Fe-LMS@H2O droplets, increasing the yields of both CH3OOH and CH3COOH threefold, reaching 1,679.6 mmol·g-1Fe·h-1 (99.9%) and 790.5 mmol·g-1Fe·h-1 (91.7%), respectively.
(C) Reaction Mechanism Study
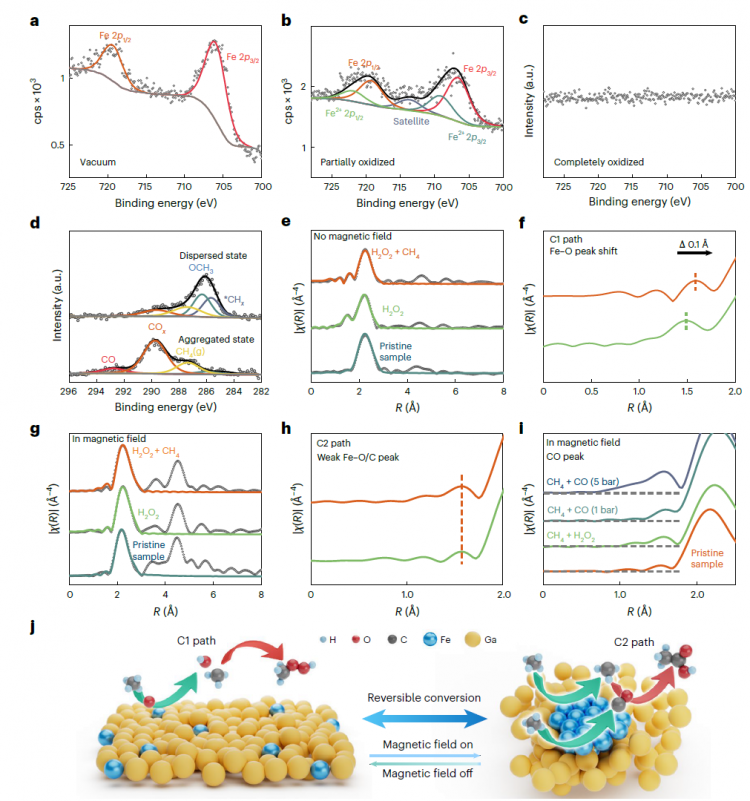
Figure 3. In-situ Experiments and Mechanism Exploration
The research team investigated the true chemical environment during the reaction process using near-ambient pressure X-ray photoelectron spectroscopy (NAP-XPS). During the reaction, both CHx (285.6 eV) and *OCH3 (286.4 eV) intermediate species were simultaneously detected on the dispersed-state catalyst. On the aggregated-state catalyst, *COx (289.8 eV) was found to be the main intermediate species. To understand the selectivity of reaction pathways, the researchers conducted ab initio molecular dynamics (AIMD) simulations and density functional theory (DFT) calculations.
In the absence of a magnetic field, two iron atoms in the liquid gallium-based alloy tend to disperse into two iron single atoms due to entropy effects. However, under magnetic fields, the two iron atoms aggregate into iron dimers. On Fe1-LMS, CH4 dehydrogenation is the rate-determining step with an energy barrier of 1.05 eV. The formed CH3O continues dehydrogenation on Fe1-LMS with a high energy barrier of 3.03 eV, which is unfavorable for forming higher oxidation state oxides. On Fe4-LMS, CH3O is converted to CO through continuous dehydrogenation steps, and then CO couples with CH3 to generate CH3CO, ultimately forming CH3COOH. The rate-determining step of this pathway (CH3CO + OH → CH3COOH) has an energy barrier of 0.95 eV.
The research team prepared an Fe-LMS catalyst with sensitive magnetic field response, which can achieve reversible conversion of the main liquid products in methane oxidation at room temperature through external magnetic field control. The external magnetic field manipulates the spin orientation of iron single atoms, guiding the directional movement and aggregation of iron atoms from the dispersed state. Magnetic field switching affects the nanostructure, local coordination environment, and electron spin of Fe-LMS, leading to the formation of intermediate species that determine the reaction pathway. In particular, the changes in iron atom aggregation state and spin state under external magnetic field break the adsorption energy scaling relationship with reaction molecules, altering the reaction selectivity. This work provides guidance for precisely controlling catalyst reaction pathways through magnetic fields, demonstrating the potential advantages of magnetic field-based catalytic regulation in improving reaction selectivity, simplifying reaction devices, and reducing industrial synthesis costs.
Paper Link: https://www.nature.com/articles/s41565-025-02029-5

Yuen Wu
Professor and PhD Supervisor at the Department of Applied Chemistry, University of Science and Technology of China (USTC), concurrently a part-time professor at the First Affiliated Hospital of USTC, and a recipient of the Chang Jiang Distinguished Professor title from the Ministry of Education. In recent years, Professor Wu has focused on the rational design of single-atom and cluster catalysts and their exploration in industrial applications, achieving remarkable accomplishments in the precise activation of "chemical bonds" in small molecules in the fields of energy and catalysis. In 2015, he received funding from the National Natural Science Foundation of China's Excellent Young Scientists Fund; in 2017, he was funded by the National Key R&D Program of China's Nanotechnology Special Youth Project and served as Principal Investigator; in 2018, he received the Chinese Chemical Society's Nanochemistry Rising Star Award; in 2019, he received the Chinese Chemical Society's Young Chemist Award; in 2020, he received the Fok Ying Tung Young Teacher Award; in 2023, he received the Optics Valley Achievement Transformation Award; and in 2025, he received the Chinese Chemical Society-Royal Society of Chemistry Young Chemist Award.
In recent years, as corresponding author, he has published more than 170 academic papers in mainstream international journals, including Nat. Mach. Intell.(1), Nat. Nanotechnol.(1), Nat. Catal.(3), Nat. Synth.(1), PNAS(2), J. Am. Chem. Soc.(16), Angew. Chem. Int. Ed.(20), Nat. Commun.(14), Adv. Mater.(10), Joule(2), etc. His papers have been cited more than 33,000 times, with an h-index of 85. He was selected as a Clarivate Highly Cited Researcher from 2020 to 2024. He currently serves as an Editorial Board Member for the journal Industrial Chemistry & Materials, an Editorial Board Member for Science China Materials, a Guest Editor for Small Methods (Single-Atom Catalysis Special Issue), a Youth Editorial Board Member for the Chinese Journal of Inorganic Chemistry, a Youth Editorial Board Member for Chemical Research in Chinese Universities, a Committee Member of the Fuel Cell Branch of the Internal Combustion Engine Association, a Committee Member of the CO2 Division of the Chinese Chemical Society, and a Committee Member of the Nanozyme Division of the Chinese Chemical Society.
中科大团队Nature Nanotechnology
磁场调控的Fe嵌入液态金属催化剂实现甲烷氧化选择性切换
一、研究背景:传统催化剂的局限与液态金属的新机遇
传统的甲烷氧化催化剂主要基于固体材料,其中活性位点的化学环境决定了催化剂的活性和选择性。单原子催化剂通常只产生C1产物,而具有多个原子位点的团簇或颗粒通常产生C2或更长链的碳氢化合物。然而,这些固体催化剂有一个固有的缺陷:其刚性固体结构限制了在反应过程中修改活性位点化学构型的能力,这显著限制了调整催化活性和选择性的可能性。
液态金属作为新兴材料,为开发甲烷氧化催化剂提供了一种新方法。其独特的物理和化学性质,如低熔点、高导热性和优异的化学稳定性,使其成为理想的催化剂载体。与固体催化剂相比,液态金属在室温下保持液态,原子不受晶格限制,从而提供了动态可调结构的可能性。与传统的催化调节方法不同,磁场很容易由永磁体产生,并允许在较温和的条件下外部控制催化反应。
二、研究内容:Fe-LMS催化剂的设计与性能验证
(一)催化剂合成与结构表征
图1.Fe-LMS结构表征
研究团队设计了一种嵌入铁的液态金属催化剂(Fe-LMS),以共晶合金galinstan作为弱结合基底。这种Fe-LMS催化剂对外部磁场强度变化产生响应,实现铁原子在孤立单原子(Fe₁-LMS)和聚集团簇(Feₙ-LMS)之间的可逆快速重新排列。
通过高角环形暗场扫描透射电子显微镜(HAADF-STEM)、扫描电子显微镜(SEM)、能量色散X射线光谱(EDS)、同步辐射X射线3D成像重构等先进表征技术,研究人员对催化剂的结构进行了深入分析。在0 G磁场条件下,样品显示铁/镓铟锡合金元素的非晶态分散。相反,500 G磁场会诱导α-Fe(100)团簇形成(晶格间距0.206 nm),而镓保持均匀分布。X射线吸收精细结构(XAFS)分析揭示了磁场诱导的结构重组。
(二)磁场调控的甲烷氧化反应
图2.甲烷氧化催化性能
甲烷氧化反应在高压高压釜中使用Fe-LMS液滴催化剂和稀释的H2O2进行。在没有磁场的情况下,Fe-LMS主要产生CH₃OOH(471.1 mmol·g⁻¹_Fe·h⁻¹,94.5%)。在500 G下,产物选择性逆转,CH₃COOH成为主要产物(230.6 mmol·g⁻¹_Fe·h⁻¹,91.9%)。超声分散产生100 nm的Fe-LMS@H2O液滴,使CH₃OOH和CH₃COOH的产率都增加了三倍,分别达到1,679.6 mmol·g⁻¹_Fe·h⁻¹(99.9%)和790.5 mmol·g⁻¹_Fe·h⁻¹(91.7%)。
(三)反应机理研究
图3.原位实验与机理探索
研究团队通过近常压X射线光电子能谱(NAP-XPS)研究反应过程中的真实化学环境。在反应过程中,呈分散状态催化剂上同时检测到CHₓ(285.6 eV)和*OCH₃(286.4 eV)中间体物种。在聚集状态催化剂上,*COx(289.8 eV)被发现为主要中间体物种。为了理解反应通路的选择性,研究人员进行了从头算分子动力学(AIMD)模拟和密度泛函理论(DFT)计算。在无磁场时,液态镓基合金中的两个铁原子由于熵效应倾向于分散为两个铁单原子。然而,在磁场下,两个铁原子聚集成铁二聚体。在Fe₁-LMS上,CH₄脱氢是速率决定步,能垒为1.05 eV。形成的CH₃O在Fe₁-LMS上继续脱氢的能垒高达3.03 eV,不利于形成更高氧化态的氧化物。在Fe₄-LMS上,CH₃O通过连续脱氢步骤转化为CO,然后CO与CH₃耦合生成CH₃CO,最终形成CH₃COOH。该路径的速率决定步(CH₃CO + OH → CH₃COOH)能垒为0.95 eV。
研究团队制备了一种具有灵敏磁场响应的Fe-LMS催化剂,它能够在室温下通过外部磁场控制实现甲烷氧化主要液体产物的可逆转化。外部磁场操控铁单原子的自旋取向,引导铁原子从分散态定向移动和聚集。磁场切换影响Fe-LMS的纳米结构、局部配位环境和电子自旋,导致产生决定反应路径的中间物种。特别是,外部磁场存在下铁原子聚集态和自旋态的变化打破了与反应分子的吸附能标度关系,改变了反应选择性。这项工作为通过磁场精确控制催化剂反应路径提供了指导,展示了基于磁场的催化调节在提高反应选择性、简化反应装置和降低工业合成成本方面的潜在优势。
论文链接:https://www.nature.com/articles/s41565-025-02029-5
吴宇恩,中国科学技术大学应用化学系教授及博士生导师,同时担任中国科学技术大学第一附属医院的兼职教授,并被授予教育部长江特聘教授称号。近年来,吴教授专注于单原子和团簇催化剂的理性设计,以及这些催化剂在工业应用中的探索,特别是在能源和催化领域小分子“化学键”的精准活化方面取得了显著成就。2015年获基金委优秀青年基金资助,2017年获国家重点研发计划纳米专项青年项目资助并任首席,2018年获得中国化学会纳米化学新锐奖,2019年获得中国化学会青年化学奖,2020年获得霍英东青年教师奖,2023年获得光谷成果转化奖,2025年获得中国化学会−英国皇家化学会青年化学奖。近年来,以通讯作者在国际主流期刊发表学术论文170余篇,包含 Nat. Mach. Intell.(1), Nat. Nanotechnol.(1), Nat.Catal.(3), Nat. Synth. (1), PNAS (2), J. Am. Chem. Soc.(16), Angew.Chem. Int. Ed. (20), Nat. Commun. (14), Adv. Mater. (10) Joule (2)等,论文总引用33000余次,h-index 85, 2020-2024年入选科睿唯安高被引科学家。目前担任期刊Industrial Chemistry & Materials编委,Science China Materials编委,Small Methods客座编辑(单原子催化专刊),无机化学学报青年编委,Chemical Research in Chinese Universities青年编委,内燃机协会燃料电池分会委员以及中国化学会CO2分会委员,中国化学会纳米酶分会委员。


